MYFData
FDA Guideline FDA Alerts FDA Trends Knowledge Base with FSMA TRACKER
The FSMA TRACKER™ powered by MYFDATA™ platforms eliminate wasted hours searching FDA’s website, reducing employee costs for Quality Assurance while, simultaneously, providing better FDA compliance with strict FDA guidelines.
- FDA API
- FEI API
- IRES
- FEI Portal



![]() Trusted by 1000+ companies, importers, and compliance professionals who rely on FDA data every day.
Trusted by 1000+ companies, importers, and compliance professionals who rely on FDA data every day.
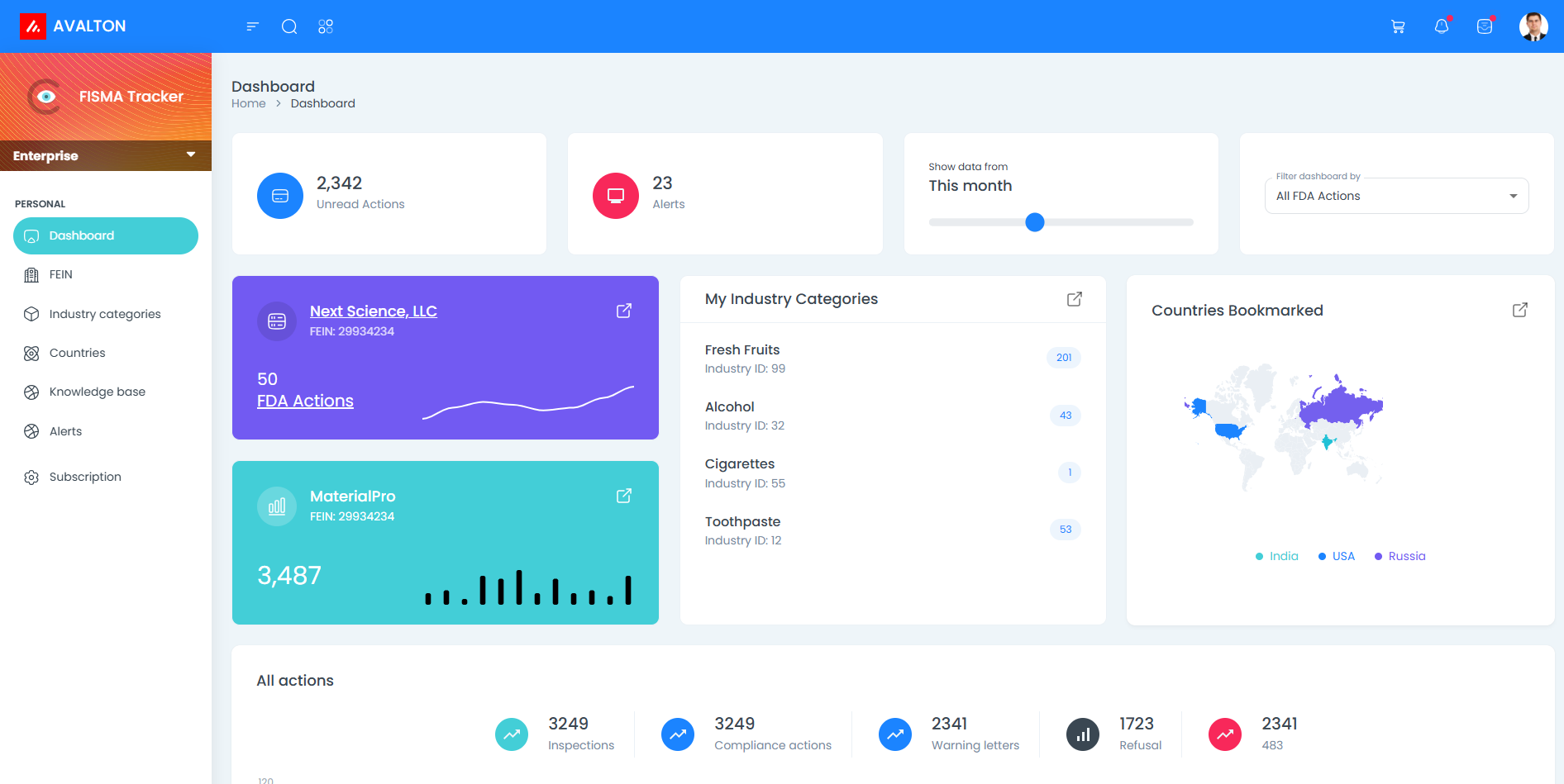
Regulatory Monitoring Capability
FSMA Tracker allow customers to monitor the FDA compliance status of U.S. importers and exporters involved with FDA-regulated .
- No Action Indicated (NAI)
- Voluntary Action Indicated (VAI)
- Official Action Indicated(OAI)
- Inspection citations
- Forms 483
Inspections
To evaluate whether a manufacturer is complying with quality manufacturing practices.
into FDA recalls, helping you track affected products and respond quickly to compliance risks
Recalls
Stay ahead of potential disruptions with reliable recall data that supports faster decisions and stronger compliance.
- Warning Letters
- Closeout Letters
- Response Letters
- Injunctions
- Seizures
Compliance Actions
Enables organizations to track, manage, and respond to FDA warning letters, recalls, and enforcement actions in real time.
- Import Alerts
- Import Refusals
- Import Detentions
- Release with Comment
- Voluntary Destruction
- Re-export
Imports
Visibility into FDA import alerts and restrictions, helping organizations monitor compliance and manage affected products at the border.
-
Find your FEIN1
-
Configure2
-
Receive the required information3
Supply Chain Insight
Easily track importers, exporters, and manufacturers. Follow the companies you care about and get instant updates whenever new FDA actions are issued.
Track Countries with FSMA Tracker
The platform allows users to monitor FDA compliance activities by Country of origin.
Monitor Compliance Actions with FDA Integrations
Seamlessly access recalls, inspections, import alerts, and enforcement data — all in one place, updated in real time.



Dynamic Templates & Customization
Real Stories, Real Results
Real success stories from businesses using our AI copywriting tool to scale faster and convert better.
Absolutely incredible! The AI-generated content is high-quality, engaging, and feels natural.
Seriously, a total time-saver! ⏳🔥
❤️❤️❤️
Ashley Wilson
The ability to instantly generate multiple variations of ad copy! I can A/B test effortlessly and optimize conversions.
Feels like having a copywriting team on demand! 🎯💡
Bobby Shell
The ability to instantly generate multiple variations of ad copy! I can A/B test effortlessly and optimize conversions.
Feels like having a copywriting team on demand! 🎯💡
Bobby Shell
Flexible Plans for Every Creator
Start for free. Upgrade anytime. No hidden fees.
Best for personal use, students
- Unlimited users
- 3 Maximum tracked Supply Chain Actors (FEIN)
- 5 Maximum tracked Industry Categories
- 1 Maximum tracked Countries
- 1 month of historical compliance data
Best for small businesses
- Unlimited users
- 25 Maximum tracked Supply Chain Actors (FEIN)
- 10 Maximum tracked Industry Categories
- 5 Maximum tracked Countries
- 12 months of historical compliance data
- New FDA Actions Alerts
Best for large businesses
- Unlimited users
- 100 Maximum tracked Supply Chain Actors (FEIN)
- ALL Maximum tracked Industry Categories
- 10 Maximum tracked Countries
- 5 years of historical compliance data
- New FDA Actions Alerts
- Analytics and trends alerts
- Knowledge base
Best for personal use, students
- Unlimited users
- 3 Maximum tracked Supply Chain Actors (FEIN)
- 5 Maximum tracked Industry Categories
- 1 Maximum tracked Countries
- 1 month of historical compliance data
Best for small businesses
- Unlimited users
- 25 Maximum tracked Supply Chain Actors (FEIN)
- 10 Maximum tracked Industry Categories
- 5 Maximum tracked Countries
- 12 months of historical compliance data
- New FDA Actions Alerts
Best for large businesses
- Unlimited users
- 100 Maximum tracked Supply Chain Actors (FEIN)
- ALL Maximum tracked Industry Categories
- 10 Maximum tracked Countries
- 5 years of historical compliance data
- New FDA Actions Alerts
- Analytics and trends alerts
- Knowledge base
